 Date: 2018-08-17 14:46:13Clinical Laboratory Improvement Amendments Healthcare in the United States Health Quality management Medical laboratory Verification and validation Laboratory Evaluation | |  Centers for Medicare & Medicaid Services, HHS from the clinic or center and the conditions for release of information. (3) The patient’s written consent is required for release of information not authorized to be relea Centers for Medicare & Medicaid Services, HHS from the clinic or center and the conditions for release of information. (3) The patient’s written consent is required for release of information not authorized to be relea
Add to Reading ListSource URL: www.gpo.govDownload Document from Source Website File Size: 602,49 KBShare Document on Facebook
|


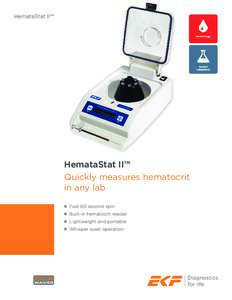


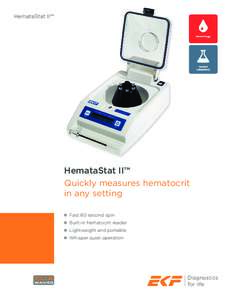
 Centers for Medicare & Medicaid Services, HHS from the clinic or center and the conditions for release of information. (3) The patient’s written consent is required for release of information not authorized to be relea
Centers for Medicare & Medicaid Services, HHS from the clinic or center and the conditions for release of information. (3) The patient’s written consent is required for release of information not authorized to be relea