Back to Results
| First Page | Meta Content | |
|---|---|---|
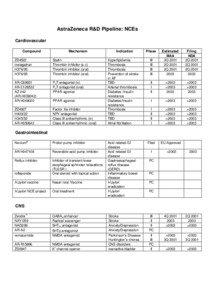 | Document Date: 2013-10-05 07:06:42Open Document Share Result on FacebookCompanyAstraZeneca / /EventFDA Phase / /FacilityAstraZeneca R&D Pipeline / Infection II AstraZeneca R&D Pipeline / /IndustryTermPrevention of stroke / /MedicalConditionpositive infections / CNS disorders / GERD / NSAID derivative Delta opioid NMDA Antagonist Acute /chronic pain Analgesia Acute /chronic / Filed TBD COPD III 3Q / Acute /chronic pain Acute /chronic / II antagonist Hypertension / TBD Asthma/COPD I 1Q / stroke / Premenopausal Adjuvant Breast Cancer III 4Q / reflux disease / Neutropenics Cystic Fibrosis III 3Q / Parkinson’s Disease Huntington’s chorea CNS / /MedicalTreatmentrelaxations / /NaturalFeatureNa channel / /OrganizationEuropean Union / /PersonAsthma Asthma III II / Paediatrics Asthma III III / /Positioncandidate for licensing activity AR-R15896 / /ProductSeroquel / Zomig / Pulmicort / glycine / Accolate / Merrem / Naropin / Nexium / Iressa / naproxen / Arimidex / Symbicort / Faslodex / Atacand / Zestril / Plendil / Casodex / Zoladex / Respiratory Symbicort ®Turbuhaler / Hypertension Atacand® Plus Phase Estimated / /ProvinceOrStateNew Brunswick / /SocialTag |