<--- Back to Details
| First Page | Document Content | |
|---|---|---|
 Date: 2015-07-02 05:24:35Health European Medicines Agency Summary of Product Characteristics MedDRA Adverse drug reaction Adverse effect Pseudoephedrine Pharmacovigilance Pharmacology Pharmaceutical sciences Medicine |
Add to Reading List |
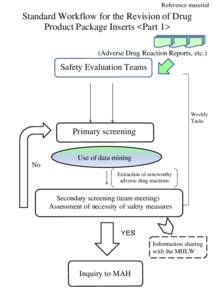 | Reference material Standard Workflow for the Revision of Drug Product Package Inserts
|
 | Managing patients’ safety: Need for PharmacoVigilance Dr. Hanmant Barkate, MBBS, MD Associate Vice-President& Head Medical, Clinical & Regulatory affairs Intas Pharmaceuticals Ltd., AhmedabadDocID: 1rcEG - View Document |
 | PHARMACOVIGILANCE GUIDE FOR ADVERSE DRUG REACTION MONITORING AND CAUSALITY ASSESSMENT, 2015 Post marketing Control Division,DocID: 1p9e5 - View Document |
 | REVIEW ARTICLE A New, Evidence-based Estimate of Patient Harms Associated with Hospital Care John T. James, PhDDocID: 1p3Uj - View Document |
 | Pharmacovigilance: Monitoring the Safety & Efficacy of Animal Medicines Summary This documentDocID: 1oBM7 - View Document |
 Text of introduction - January 2013
Text of introduction - January 2013