<--- Back to Details
| First Page | Document Content | |
|---|---|---|
 Health Tobacco Food and Drug Administration Smoking Electronic cigarettes Tobacco control Smoking cessation Tobacco in the United States Center for Tobacco Products Federal Food Drug and Cosmetic Act Cigarette Premarket tobacco application |
Add to Reading List |
 | September 12, 2018 Japan Tobacco International, USA, Inc. Glenpointe Centre West 500 Frank W. Burr Blvd. #24 Teaneck, NJDear Mr. Jerry Loftin:DocID: 1xVgy - View Document |
 | Notice to Industry: Additional Tobacco Products Now Regulated by the Food and Drug AdministrationDocID: 1xVey - View Document |
 | Tobacco Region Revitalization Commission for the year ended June 30, 2017DocID: 1xUYe - View Document |
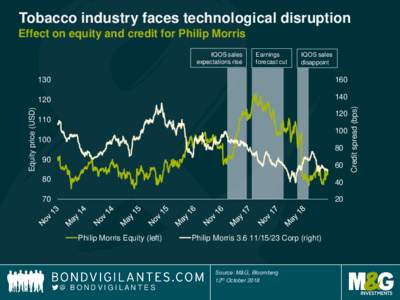 | Tobacco industry faces technological disruption Effect on equity and credit for Philip Morris Earnings forecast cut IQOS salesDocID: 1xUsE - View Document |
 | Illicit Trade in Tobacco Products after Implementation of an FDA Product StandardDocID: 1xTPY - View Document |
 September 12, 2018 Japan Tobacco International, USA, Inc. Glenpointe Centre West 500 Frank W. Burr Blvd. #24 Teaneck, NJDear Mr. Jerry Loftin:
September 12, 2018 Japan Tobacco International, USA, Inc. Glenpointe Centre West 500 Frank W. Burr Blvd. #24 Teaneck, NJDear Mr. Jerry Loftin: