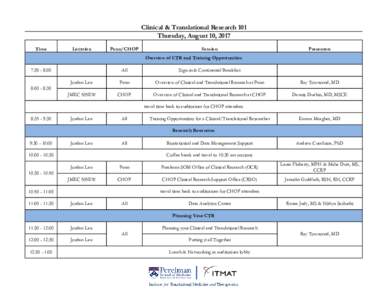
<--- Back to Details
| First Page | Document Content | |
|---|---|---|
 Clinical research Center for Devices and Radiological Health Center for Drug Evaluation and Research Biologic Center for Biologics Evaluation and Research Surrogate endpoint FDA Fast Track Development Program Clinical trial Federal Food Drug and Cosmetic Act Food and Drug Administration Medicine Health |
Add to Reading List |
 Regulatory Basis for U.S. Drug & Biologics Approval Paul G. Kluetz, M.D. Office of Hematology and Oncology Products
Regulatory Basis for U.S. Drug & Biologics Approval Paul G. Kluetz, M.D. Office of Hematology and Oncology Products