1 | Add to Reading ListSource URL: www.forefrontmed.comLanguage: English - Date: 2014-05-29 13:16:27
|
|---|
2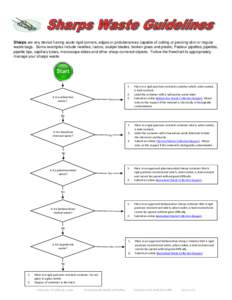 | Add to Reading ListSource URL: www.ehs.uci.eduLanguage: English - Date: 2016-02-02 15:20:53
|
|---|
3 | Add to Reading ListSource URL: www.hunton.comLanguage: English - Date: 2016-05-18 11:22:36
|
|---|
4 | Add to Reading ListSource URL: www.horiba.comLanguage: English - Date: 2016-01-14 03:36:17
|
|---|
5 | Add to Reading ListSource URL: www.r-project.orgLanguage: English - Date: 2015-03-04 04:31:16
|
|---|
6 | Add to Reading ListSource URL: airgasdryice.netLanguage: English |
|---|
7 | Add to Reading ListSource URL: www.fp6-creation.ruLanguage: English - Date: 2009-10-01 20:14:10
|
|---|
8 | Add to Reading ListSource URL: www.fp6-creation.ruLanguage: English - Date: 2009-10-01 20:13:41
|
|---|
9 | Add to Reading ListSource URL: www.saitama-j.or.jpLanguage: English - Date: 2013-02-18 03:02:25
|
|---|
10 | Add to Reading ListSource URL: www.cnsc-ccsn.gc.caLanguage: English - Date: 2015-03-10 16:28:05
|
|---|